France opens up Covid vaccinations for all 5 to 11 year-olds
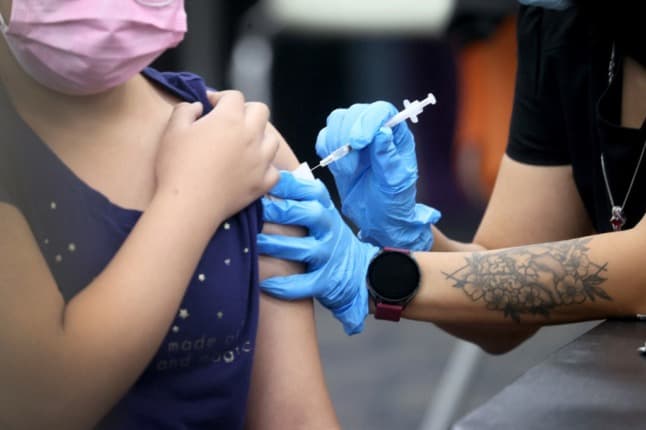
France on Wednesday opened up the Covid vaccination programme to children aged between 5 and 11, here's how it works.
When?
France was already offering the vaccine to 5-11 year-olds who were in a high risk group because of a medical condition, but on Wednesday December 22nd this opens up to all children in this age group.
Health minister Olivier Véran, speaking on Wednesday morning, said: "It's official, today we're opening up vaccination for children".
He added that online booking platforms will be updated "shortly" to include the 5-11 age group, and that children can be given the shot in vaccine centres or by GPs or family doctors.
For younger children at least one parent or legal guardian must be present at the appointment, and they must sign a consent form - you can find that HERE.
Children in the 12-18 age group have been eligible for the vaccine since the summer.
READ ALSO ‘Take a test’: The official Covid recommendations for Christmas in France
What do the experts say?
The Haute Autorité de santé (HAS) gave the green light to vaccinate all children aged 5-11 on Monday. It noted however that the vaccination of children should not be made compulsory.
Last week, the Comité consultatif national d’éthique said that it also supported the vaccination of this age group, on a voluntary basis.
Which vaccine will children receive?
The European Medicines Agency approved the Pfizer vaccine for children aged five to 11 in November. It is the only vaccine to have EU approval for this age group.
Moderna is in phase three of its clinical trial but the first results are promising and an authorisation request has been made - but France has set a lower age-limit on Moderna and AstraZeneca vaccines.
How can you make an appointment?
You can make appointments in the usual ways, either direct with your doctor or online using services such as Doctolib or Vite Ma Dose, which the minister says will be updated "shortly" to include the younger age group category.
READ ALSO How to get a Covid-19 booster dose appointment in France
Will children receive the same dose as adults?
No. The dose authorised for children by the EMA and HAS is 10 microgrammes, compared to 30 microgrammes in doses for adults.
‼️ EMA recommends approval of BioNTech/Pfizer’s #COVID19vaccine, Comirnaty, for children aged 5 to 11.
In this population, the dose of #Comirnaty will be lower than that used in people aged 12 and above.
Read the full press release: https://t.co/uqqBAklVvl pic.twitter.com/NZQhli4SDl
— EU Medicines Agency (@EMA_News) November 25, 2021
The HAS has recommended that the two vaccine doses are given 21 days apart.
Is vaccination against Covid-19 compulsory for children of this age?
No, vaccination is voluntary as it is for adults. And formal parental consent will be required. So, expect a form if you do decide to vaccinate your child.
Okay, so it’s not compulsory - but will health pass rules be extended to include five to 11 year olds?
The government has said children in this age group will not require a health pass, so - as it stands - those under 12 will not be required to show a pass sanitaire to go to cafes and restaurants, or attend, for example, sporting events.
READ ALSO France sets 7 month limit on Covid health pass and opens up booster jabs to all adults
Could they be barred from school if parents decide not to get them vaccinated?
No. Children aged 12 to 17 who have not been vaccinated still go to school as normal although they are subject to stricter rules around testing and self-isolation if there is an outbreak in class.
READ ALSO Covid-19 in French schools: What are the rule changes?
And what about 12-17 year olds? Why aren't they being called back for a booster dose?
France's Direction générale de la Santé said on Tuesday, December 7th: “To date, only people aged 12 to 17 with co-morbidities or who are immunocompromised are eligible for the vaccination booster five months after their last vaccine injection or their last infection. Immunocompetent adolescents are not yet affected by the recall.
“The data we currently have show more stable protection against infection in those under 18 years of age than in adults. We are awaiting additional data and the opinion of the scientific authorities before considering opening the recall campaign to these people.”
Comments
See Also
When?
France was already offering the vaccine to 5-11 year-olds who were in a high risk group because of a medical condition, but on Wednesday December 22nd this opens up to all children in this age group.
Health minister Olivier Véran, speaking on Wednesday morning, said: "It's official, today we're opening up vaccination for children".
He added that online booking platforms will be updated "shortly" to include the 5-11 age group, and that children can be given the shot in vaccine centres or by GPs or family doctors.
For younger children at least one parent or legal guardian must be present at the appointment, and they must sign a consent form - you can find that HERE.
Children in the 12-18 age group have been eligible for the vaccine since the summer.
READ ALSO ‘Take a test’: The official Covid recommendations for Christmas in France
What do the experts say?
The Haute Autorité de santé (HAS) gave the green light to vaccinate all children aged 5-11 on Monday. It noted however that the vaccination of children should not be made compulsory.
Last week, the Comité consultatif national d’éthique said that it also supported the vaccination of this age group, on a voluntary basis.
Which vaccine will children receive?
The European Medicines Agency approved the Pfizer vaccine for children aged five to 11 in November. It is the only vaccine to have EU approval for this age group.
Moderna is in phase three of its clinical trial but the first results are promising and an authorisation request has been made - but France has set a lower age-limit on Moderna and AstraZeneca vaccines.
How can you make an appointment?
You can make appointments in the usual ways, either direct with your doctor or online using services such as Doctolib or Vite Ma Dose, which the minister says will be updated "shortly" to include the younger age group category.
READ ALSO How to get a Covid-19 booster dose appointment in France
Will children receive the same dose as adults?
No. The dose authorised for children by the EMA and HAS is 10 microgrammes, compared to 30 microgrammes in doses for adults.
‼️ EMA recommends approval of BioNTech/Pfizer’s #COVID19vaccine, Comirnaty, for children aged 5 to 11.
— EU Medicines Agency (@EMA_News) November 25, 2021
In this population, the dose of #Comirnaty will be lower than that used in people aged 12 and above.
Read the full press release: https://t.co/uqqBAklVvl pic.twitter.com/NZQhli4SDl
The HAS has recommended that the two vaccine doses are given 21 days apart.
Is vaccination against Covid-19 compulsory for children of this age?
No, vaccination is voluntary as it is for adults. And formal parental consent will be required. So, expect a form if you do decide to vaccinate your child.
Okay, so it’s not compulsory - but will health pass rules be extended to include five to 11 year olds?
The government has said children in this age group will not require a health pass, so - as it stands - those under 12 will not be required to show a pass sanitaire to go to cafes and restaurants, or attend, for example, sporting events.
READ ALSO France sets 7 month limit on Covid health pass and opens up booster jabs to all adults
Could they be barred from school if parents decide not to get them vaccinated?
No. Children aged 12 to 17 who have not been vaccinated still go to school as normal although they are subject to stricter rules around testing and self-isolation if there is an outbreak in class.
READ ALSO Covid-19 in French schools: What are the rule changes?
And what about 12-17 year olds? Why aren't they being called back for a booster dose?
France's Direction générale de la Santé said on Tuesday, December 7th: “To date, only people aged 12 to 17 with co-morbidities or who are immunocompromised are eligible for the vaccination booster five months after their last vaccine injection or their last infection. Immunocompetent adolescents are not yet affected by the recall.
“The data we currently have show more stable protection against infection in those under 18 years of age than in adults. We are awaiting additional data and the opinion of the scientific authorities before considering opening the recall campaign to these people.”
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.