France recommends Covid-19 vaccine for 'high-risk' children from age 5
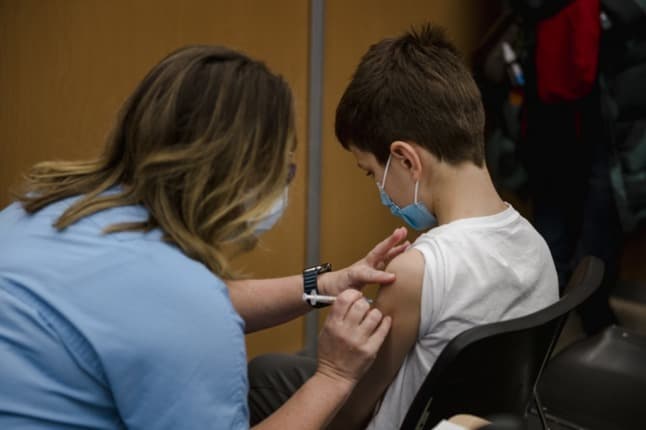
Health authorities in France have recommended that children aged five to 11 who have a ‘higher risk of developing a serious form’ of Covid-19 should be vaccinated.
Vaccination is also recommended for children who live with or are otherwise close to people who, for health reasons of their own, cannot be vaccinated.
At least 360,000 children, according to official figures, with a range of chronic illnesses - including heart disease, heart defects, serious lung disease, cancer, kidney disease, liver disease, or severe forms of asthma would be eligible, the Haute Autorité de Santé (HAS) has said.
In France the HAS is advisory and it is up to the government to set policy, but it is rare for the government to go against HAS advice.
On the subject of vaccines for all 5-11 year-olds, health minister Olivier Véran said in his speech last week that a consultation was ongoing. He said that a decision would be made on this in the New Year.
READ ALSO Reader question: Do I need a vaccine booster if I’ve already had Covid?
“Doctors may also offer this vaccination to children who have rare pathologies that are not on the list but for which there is also a risk,” Daniel Floret, vice-president of the HAS’s technical commission for vaccinations told Franceinfo.
“It is also recommended for children close to immunocompromised people because they have very limited protection, who respond poorly to vaccines, so vaccinating those around them is one way to protect them.”
A smaller dose of the Pfizer/BioNTech Covid-19 vaccine, three times lower than the dose for adults, is available for children who fall into the vulnerable category.
The recommendation comes a week after the European Medicines Agency recommended a 10 microgram dose of the Pfizer Covid-19 vaccine for all children aged between five and 11 years old.
READ ALSO France sets 7 month limit on Covid health pass and opens up booster jabs to all
The HAS has yet to publish its advice on this subject.
“It is well known that children usually have mild forms of the disease so the individual benefit of vaccination is not as obvious as in adults or in children who have risk factors,” Floret said, explaining that the HAS was waiting for further data from the US.
“We will soon have the data of the American experience since Americans will have vaccinated several million children, so if there are particular tolerance problems, it will appear in clinical trials,” he said.
Comments
See Also
Vaccination is also recommended for children who live with or are otherwise close to people who, for health reasons of their own, cannot be vaccinated.
At least 360,000 children, according to official figures, with a range of chronic illnesses - including heart disease, heart defects, serious lung disease, cancer, kidney disease, liver disease, or severe forms of asthma would be eligible, the Haute Autorité de Santé (HAS) has said.
In France the HAS is advisory and it is up to the government to set policy, but it is rare for the government to go against HAS advice.
On the subject of vaccines for all 5-11 year-olds, health minister Olivier Véran said in his speech last week that a consultation was ongoing. He said that a decision would be made on this in the New Year.
READ ALSO Reader question: Do I need a vaccine booster if I’ve already had Covid?
“Doctors may also offer this vaccination to children who have rare pathologies that are not on the list but for which there is also a risk,” Daniel Floret, vice-president of the HAS’s technical commission for vaccinations told Franceinfo.
“It is also recommended for children close to immunocompromised people because they have very limited protection, who respond poorly to vaccines, so vaccinating those around them is one way to protect them.”
A smaller dose of the Pfizer/BioNTech Covid-19 vaccine, three times lower than the dose for adults, is available for children who fall into the vulnerable category.
The recommendation comes a week after the European Medicines Agency recommended a 10 microgram dose of the Pfizer Covid-19 vaccine for all children aged between five and 11 years old.
READ ALSO France sets 7 month limit on Covid health pass and opens up booster jabs to all
The HAS has yet to publish its advice on this subject.
“It is well known that children usually have mild forms of the disease so the individual benefit of vaccination is not as obvious as in adults or in children who have risk factors,” Floret said, explaining that the HAS was waiting for further data from the US.
“We will soon have the data of the American experience since Americans will have vaccinated several million children, so if there are particular tolerance problems, it will appear in clinical trials,” he said.
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.